Thank you for visiting our Pharmacy,
Existing pharmacy customers please login to show our products.
New customers, please Register for a pharmacy account.
Free shipping on all UK orders over £50
Special offers
Regular sales and discounts
Free Next Business Day Delivery in the UK
On orders before 3pm (Monday - Friday).
Exclusive Lip Booster
Get 'QUENCH for £10!
Thank you for visiting our Pharmacy,
Existing pharmacy customers please login to show our products.
New customers, please Register for a pharmacy account.
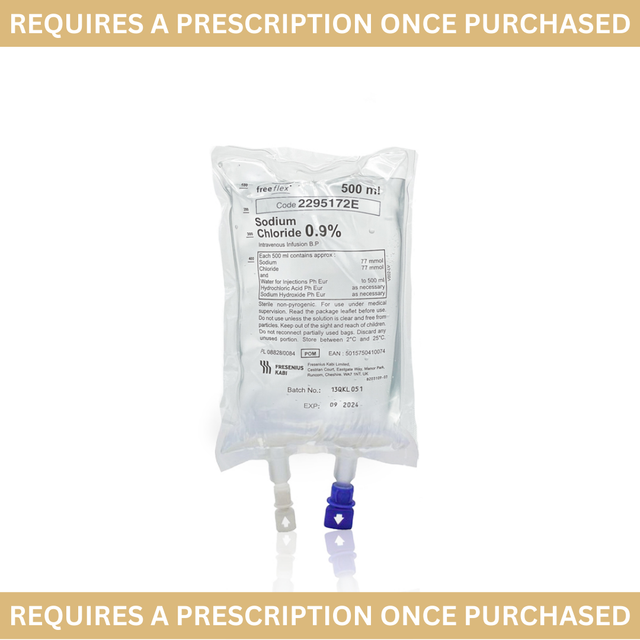
Solution for infusion - clear solution, free from visible particles.
Therapeutic indications:
Sodium Chloride 0.9% intravenous infusion is indicated for:
• Treatment of isotonic extracellular dehydration
• Treatment of sodium depletion
• Vehicle or diluent of compatible drugs for parenteral administration.
Features
Nature and contents of container:
• Bag sizes: 50, 100, 250, 500 or 1000 mL
• The bags known as Viaflo are composed of polyolefin/polyamide co-extruded plastic (PL-2442).
• The bags are overwrapped with a protective plastic pouch composed of polyamide/polypropylene.
Special precautions for storage:
• 50 and 100 ml bags: Do not store above 30°C.
• 250, 500 and 1000 ml bags: This medicinal product does not require any special storage conditions.
Shelf life:
• Chemical and physical stability of any additive at the pH of Sodium Chloride 0.9% Intravenous Infusion in the Viaflo container should be established prior to use.
• From a microbiological point of view, the diluted product must be used immediately unless dilution has taken place in controlled and validated aseptic conditions. If not used immediately, in-use storage times and conditions are the responsibility of the user.
Please note brands may vary depending on bag size.
Size
Enjoy 15% off your first order when you sign up to our mailing list.
Visit our support center
Expert help & advice
Check your order status
Updates & tracking
Returns & exchanges
All you need to know
Worldwide Shipping
UK, Europe, US & Auz